Category :
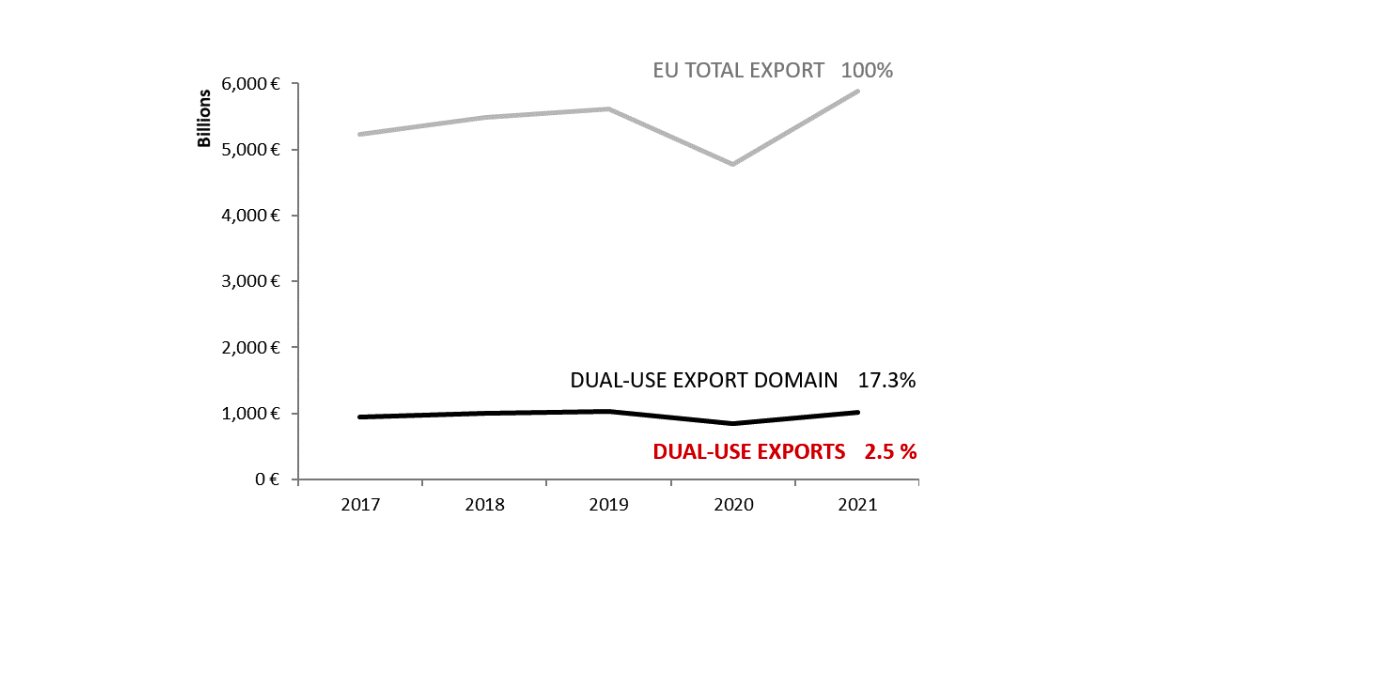
2021 EU dual-use export control statistical update On 19th October 2023, the European Commission published the statistical update[1] on the implementation of the EU Export Control Regulation of dual use items (Regulation (EU) 2021/821)[2]. This update is released, as […]
Category :
On 25 May 2023, it has been published on the Official Journal of the EU the exceptional amendments to Annex I of Regulation (EU) 2021/821 to implement the changes in the control list adopted by the non-proliferation regime Australia Group at its Plenary in July 2022.
Category :
On 11 January 2023, it has been published on the Official Journal of the EU the updated version of Annex I to EU Dual-Use Regulation (also known as EU Dual-Use Control List): Commission Delegated Regulation (EU) 2023/66 of 21 October 2022 amending Regulation (EU) 2021/821 of the European Parliament and of the Council as regards the list of dual-use items.
Category :
Report on the implementation of the new EU dual-use Regulation On 2 September 2022, the Report on the implementation of the new EU dual-use Regulation 2021/821 (hereinafter “the Regulation”) was issued.[1] The document details the implementation of the […]
Category :
As scientific research may involve sensitive subjects, it is necessary to be able to assess the extent to which research is compatible with European dual-use legislation.
We are pleased to announce the release of the new version of the dual use assistance application.
Category :
Following the eleventh gathering of the Chaudfontaine Group, in the wake of the coming into effect of the new European Dual-Use Regulation, the group of experts took the opportunity to analyse and dissect the complex EU dual-use export control regime in order to distinguish/identify the new and relevant elements which could be useful to third countries and conversely, those which are not as their raison d’être which are linked to the internal functioning of the EU.